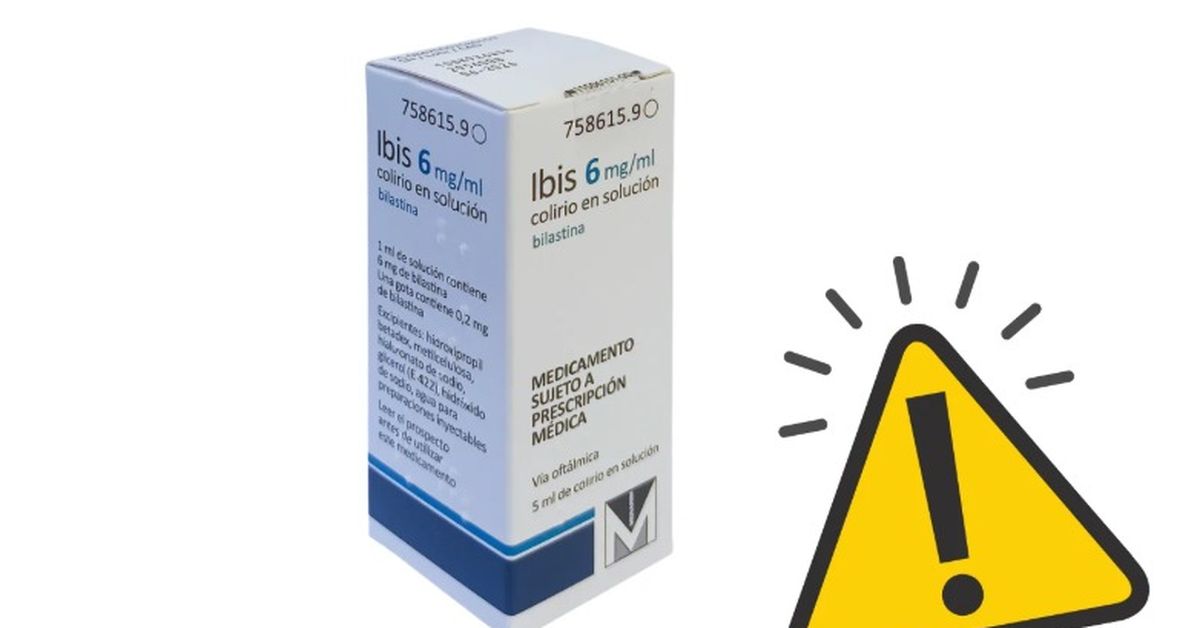
The Spanish Agency for Medicines and Health Products (Aemps) has ordered the withdrawal of several batches of Ibis 6 mg/ml eye drops in solution after obtaining an “out-of-specification result in subvisible particles of the active ingredient, detected in stability studies.” It should be noted that this quality defect does not pose a vital risk to the patient.
As Facua points out, the affected lots correspond to 2956008 (expiry date of June 30, 2026), 313160A (expiry date of January 31, 2027) and 3131908 (expiry date of December 31, 2026). This drug is manufactured by Famar Heal Th Care Services Madrid SAU and its marketing authorization holder is Menarini International Operations Luxenbourg SA.
This medication is used to treat the signs and symptoms of eye disorders that occur with seasonal allergic conjunctivitis in adults and children 2 years and older. Also to treat the signs and symptoms of eye disorders caused by an allergy to substances such as house dust mites or animal hair (perennial allergic conjunctivitis) in adults and children from 2 years of age.
The agency has classified the defect as class 2, with class 1 corresponding to a possible higher risk and class 3 being the lowest. The Aemps, dependent on the Ministry of Health, has ordered the withdrawal from the market of all distributed units of the affected batches and their return to the laboratory. Likewise, it urges the autonomous communities to monitor the withdrawal.